Nandrolone 17 Beta-Carbonates
Summary:
Researchers at the Eunice Kennedy Shriver National Institute of Child Health and Human Development are highly motivated in seeking licensing and/or collaboration partners for the development and use of androgenic compounds as contraceptives and/or hormonal therapeutics.
Description of Technology:

Safety and pharmacokinetics of oral single-dose, 28 day repeat-dose, and dose escalation study of 11-MNTDC in healthy men have been conducted. The drug was well tolerated without serious adverse events. Daily oral dose of 11β-MNTDC for 28 days in healthy men showed markedly suppressed serum gonadotropins and T concentrations without serious adverse effects. NICHD is planning another Phase I trial to test 11β-MNTDC via intramuscular injection.
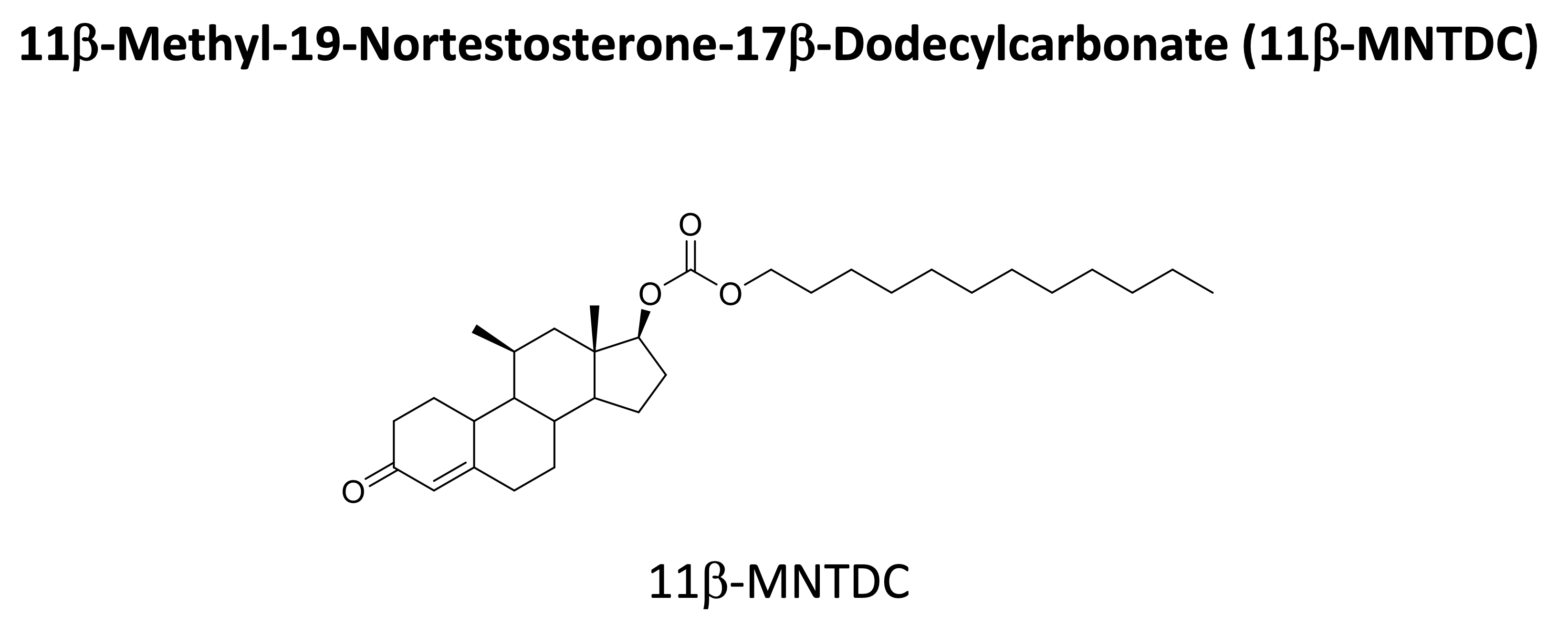
Potential Commercial Applications:
- Male contraceptive
- Treatment of hormonal diseases
Competitive Advantages:
- Significant market need for male contraceptive
- Clinical-stage asset
- No adverse events in a Phase I dose-escalation study
- Intramuscular (IM) injection
