Technology ID
TAB-1170
Spatially Selective Fixed-Optics Multicolor Fluorescence Detection System For Microfluidic Device
E-Numbers
E-223-2005-0
Lead Inventor
Morgan, Nicole
Lead IC
NIBIB
Co-Inventors
Wellner, Edward
Smith, Paul
ICs
NIBIB
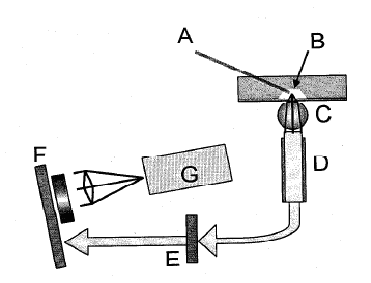
Available for licensing and commercial development is a new scheme for sensitive spatially resolved and spectrally resolved laser-induced fluorescence detection from multiple microfluidic channels. The prototype instrument has been developed and is versatile in that it contains only fixed optical parts and has simultaneous five-color detection from eight microchannels in a plastic microchip for DNA analysis. The detection scheme could be applied to fluorescence detection for any microchip-based analysis in a transparent substrate. The economies of parallel detection and the importance of spatial selectivity would make this method most useful for polymeric substrates with multiple microchannels. Free space laser excitation incident off-axis (about 60 degrees to normal on the chip) is used to minimize the coupling of laser light into the detection optical fiber. The emitted fluorescence is detected with an optical fiber-ball lens combination, one for each microchannel. The spatial selectivity is achieved by using a high refractive index 2 mm ball lens and a small-diameter (200 um) .22 NA optical fiber positioned to obtain focused light from the channel. There are no moving parts so this configuration is both more robust and more versatile than a scanning system. Furthermore, the detection optics can be freely positioned near the channel, placing minimal constraints on channel layout and design. After the emitted fluorescence is coupled into the fiber, the light is passed through a long pass filter (here, 510AELP, Omega Optics), and then spectrally dispersed using a compact imaging spectrograph (FICS, Oriel). The resulting spectra are imaged using a cooled monochrome CCD (Qimaging Retiga EXl) at 10 frames per second. This setup allows simultaneous detection of multiple dyes. The laser excitation is split into multiple spots with two cylindrical lenses and an array of spherical plano-convex lenses. The spacing of the plano-convex lenses is chosen such that the laser spots coincide with the microchannels in the chip. At each excitation spot, a ball lens and optical fiber is positioned underneath the microchannel. The other ends of the optical fiber are formed into a 1-D array and directed onto the slit of an imaging spectrograph.

Licensing Contact:
