Therapeutic to Treat Inherited Retinal Disorders and Age-Related Macular Degeneration Available for Licensing or Collaboration

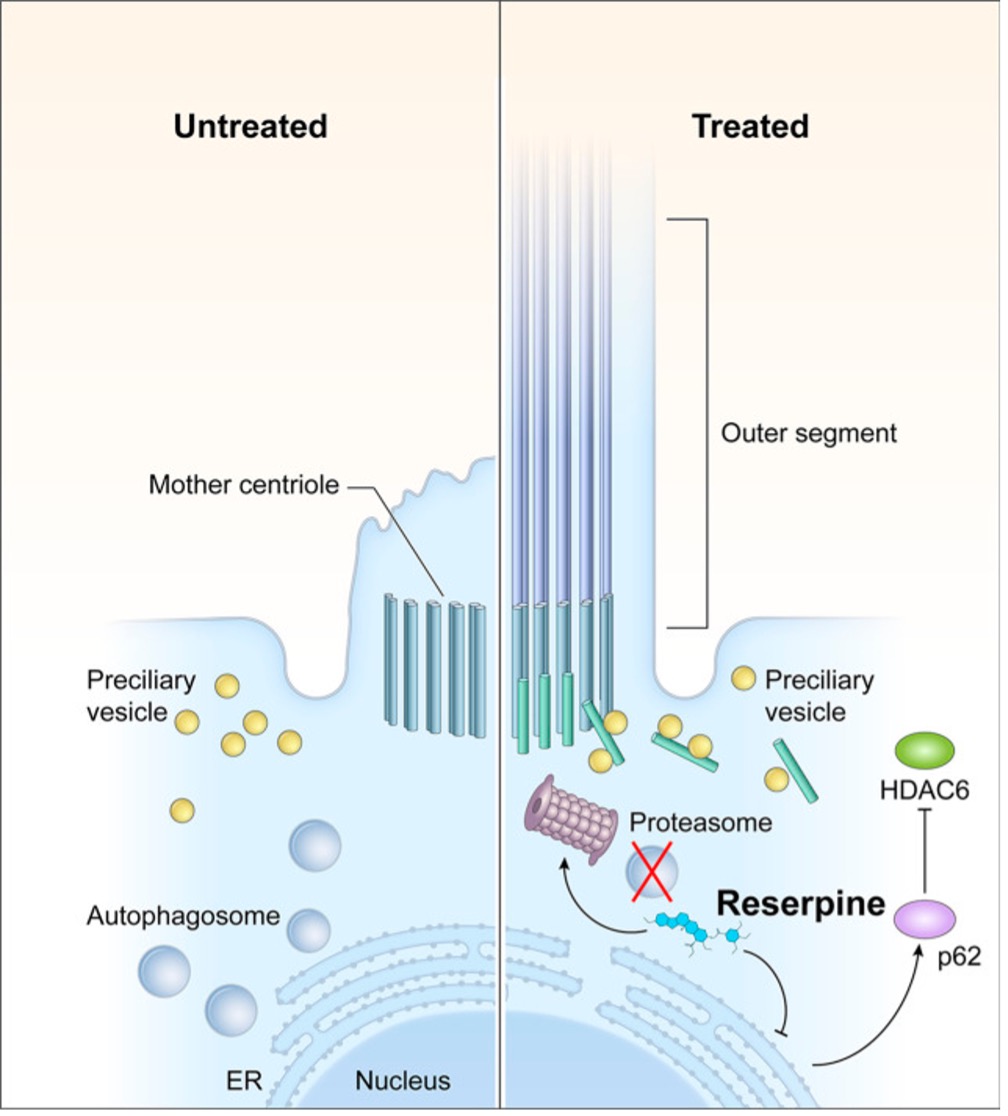
NIH inventors have developed a mutation-independent method to treat Leber congenital amaurosis (LCA), retinitis pigmentosa (RP), other inherited retinal degenerations (IRDs), and age-related macular degeneration (AMD) using the FDA-approved small molecule, Halofantrine. NIH inventors found Halofantrine helped protect photoreceptors from cell death in three different IRD model systems:1) retinal organoid model systems, 2) two different mouse models, and 3) rat models. These small molecules act by targeting several cellular pathways that are also affected in diseases of other organs, suggesting that they may be useful in treating other, mutation-independent ocular diseases. Packaging Halofantrine and other potential candidate compounds for conjunctival delivery into the eye via eye drops is under development.
There are currently no treatments for RP, which affects roughly 1 in 4000 people worldwide. There is an FDA-approved treatment for one form of LCA, however long-term data for the gene therapy achieved only transient results. AMD is the leading cause of blindness in all developed countries, and by 2040, AMD will affect approximately 288 million people worldwide.
Competitive advantages of this method include:
- Less time-consuming, costly, and labor-intensive compared to the development of individualized gene therapies
- Small molecules are scalable with cost-effective manufacturing
- Small molecules are highly penetrant with easy routes of administration
- Small molecules can be delivered via eye drops or as intra-vitreal injection
- Less intrusive and an easy administration route
- Conjunctival delivery and eye drop formulation are more accessible than retinal implant or injection
If you are interested in licensing and/or collaborating on this therapeutic to treat LCA, IRD, AMD, or other mutation-independent ocular diseases, you can view more information and contact the licensing manager via the abstract: Using FDA-approved Small Molecule Drug Reserpine and related compounds (especially Halofantrine) To Protect Photoreceptors in Inherited Retinal Degenerations and Age-Related Macular Degeneration
